Study on the selectivity of epiphytic lactic acid bacteria to the carbon sources in the medium
-
摘要:
目的 探究饲草源乳酸菌对碳源的选择性,为不同草种筛选适宜的青贮菌种。 方法 选取饲草中分离获得的16株代表性乳酸菌作为研究对象,在MRS液体培养基中以葡萄糖、蔗糖、果糖、木糖、棉籽糖以及5种糖混合为碳源,进行不同菌株发酵培养。每株乳酸菌设3个重复,每个糖处理设空白对照(不接菌)。采用高效液相色谱仪测定各处理发酵24 h后液体培养基中剩余碳源含量和乳酸产量。 结果 (1)蔗糖是大部分菌株发酵首选碳源,在棉籽糖、葡萄糖、果糖和木糖的选择上不同菌株差异较大,发酵乳杆菌(Lactobacillus fermentum,LbF-WM)首选棉籽糖和木糖,假肠膜明串珠菌(Leuconostioc pseudomesenteroides,LeP-IR)首选木糖。(2)罗伊氏乳杆菌(Lactobacillus reuteri,LbR-EG)和柠檬明串珠菌(Leuconostoc citreum,LeC-IR)对蔗糖,LbF-WM和LbR-EG对果糖, LbF-WM 、LbR-EG和LeP-IR对棉籽糖的利用率均达100%,屎肠球菌(Enterococcus faecium,EF-KG)、短乳杆菌(Lactobacillus brevis,LbB-KG)、戊糖乳杆菌(Lactobacillus pentosus,LbPe-EG)、乳酸乳球菌(Lactococcus lactis,LcL-SC)对葡萄糖的利用率高于其他碳源。(3)灰黄色肠球菌(Enterococcus durans,EG-IR)利用葡萄糖和混合糖,LbPe-EG利用果糖,肠膜明串珠菌(Leuconostoc mesenteroides,LeM-IR)利用棉籽糖和混合糖,LbPe-EG利用蔗糖,LbF-WM利用木糖,格氏乳球菌(Lactococcus garvieae,LcG-IR)利用混合糖发酵乳酸产量最高。(4)耐久肠球菌(Enterococcus durans,ED-IR)利用葡萄糖,植物乳杆菌(Lactobacillus plantarum,LbPl-IR)、LbPe-EG利用果糖, LbPl-IR和LeM-IR利用棉籽糖, LbPe-EG、LbPl-IR和ED-IR利用蔗糖, LeP- IR利用木糖,LcG-IR和EG-IR利用混合糖产乳酸效率较高。LbPl-IR在5种单糖中产酸效率均达到80%以上。 结论 不同菌株对同一碳源的利用率、产酸量和产酸效率均不同;同一株菌株对不同碳源的利用率、产酸量和产酸效率也不相同。其中,蔗糖为大部分菌株的首选碳源,蔗糖和葡萄糖的利用率和产乳酸量较高,葡萄糖的产酸效率较高;青贮调制时,可针对饲草中分布乳酸菌种类选择最适碳源。 Abstract: In order to explore the selectivity of different lactic acid bacteria (LAB) to the carbon source of medium, 16 representative LAB were isolated from forage crops as the research object were selected. In the MRS broth medium, glucose, sucrose, fructose, xylose, raffinose and mixed sugars (raffinose-glucose-fructose-sucrose-xylose) were used as carbon sources for the 16 strains. There were 3 replicates for each strain, and a blank control (without inoculation) for each sugar treatment. After 24 hours of inoculation’, the high-performance liquid chromatography was used to determine the contents of residual carbon source and lactic acid in the broth for each treatment. The results of the 16 strains showed that: (1) Sucrose was the preferred carbon source for the growth of most strains; there were differences in the selection of glucose, fructose and xylose among different strains. Lactobacillus fermentum LbF-WM preferred raffinose and xylose, and Leuconostoc pseudomesenteroides LeP-IR preferred xylose. (2) The utilization rate of sucrose reached 100% by Lactobacillus brevis LbR-EG and Leuconostoc citreum LeC-IR, Lb. fermentum LbF-WM and Lactobacillus reuteri LbR-EG used fructose, Lb. fermentum LbF-WM, Lb. reuteri LbR-EG and Le. pseudomesenteroides LeP-IR used raffinose. The utilization rate of glucose by Enterococcus faecium EF-KG, Lb. brevis LbB-KG, Lactobacillus pentosus LbPe-EG and Lactococcus lactis LcL-SC were higher than that of other carbon sources. (3) The lactic acid yield was higher by E. durans EG-IR using glucose and mixed sugar, Lb. pentosus LbPe-EG used fructose, Leuconostoc mesenteroides LeM-IR fermented raffinose and mixed sugar, Lb. pentosus LbPe-EG useed sucrose, Lb. fermentum LbF-WM used xylose, and Lactococcus garvieae LcG-IR used mixed sugar. (4) The lactic acid production was more efficient by E. durans ED-IR using glucose, Lb. plantarum LbPl-IR, Le. mesenteroides LeM-IR and Lb. pentosus LbPe-EG used fructose, Lb. plantarum LbPl-IR and Le. mesenteroides LeM-IR used raffinose, Lb. pentosus LbPe-EG, Lb. plantarum LbPl-IR and E. durans ED-IR used sucrose, and Le. pseudomesenteroides LeP-IR used xylose, Lc. garvieae LcG-IR and E. durans EG-IR used mixed sugars. The lactic acid production efficiency of Lb. plantarum LbPl-IR used 5 kinds sugars was more than 80%. In summary, the different strains had different sugar utilization, acid production and acid production efficiency in the same sugar source, and the same strain fermented different sugars also had different sugar utilization, acid production and acid production efficiency. In view of the types of LAB distributed on forage crops, selecting the most suitable carbon source would benefit to more rapid and large-scale production of lactic acid, lowering the pH value, and better preserving the nutrients of silage.-
Key words:
- epiphytic lactic acid bacteria /
- carbon source /
- glucose /
- sucrose /
- selectivity
-
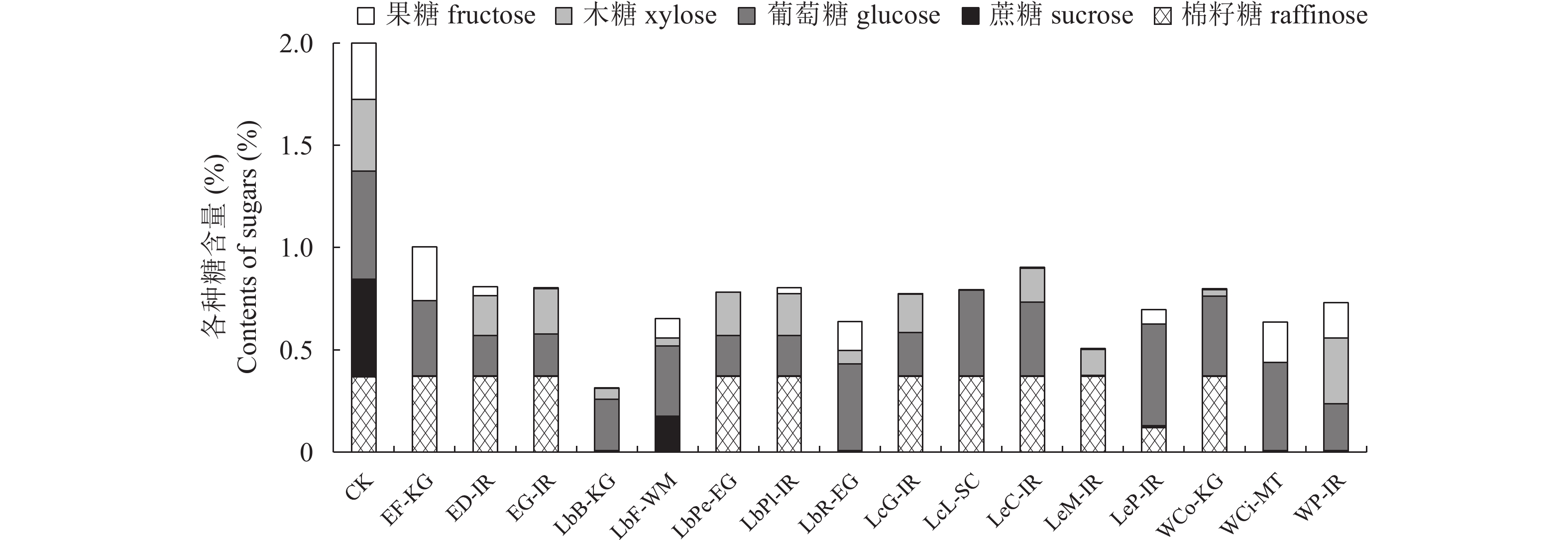
图 1 16株乳酸菌培养24 h后在5种糖混合培养基中各种糖的剩余含量
1)图中柱状数据表示混合糖培养基中剩余各糖的百分含量。2)CK,无接菌的混合糖培养基;
Figure 1. The remaining sugars content of 16 strains of LAB after 24 h cultured in mixed medium
1) The column data in the figure represents the content of various sugars remaining in the mixed sugar medium. 2) CK, mixed sugar medium without inoculation. The same as below.
表 1 16株乳酸菌的名称及来源
Table 1. The name and source of 16 LAB strains
序号
No.命名
Name种名
Species来源
Source序号
No.命名
Name种名
Species来源
Source1 EF-KG 屎肠球菌 E. faecium 热研四号王草 9 LcG-IR 格氏乳球菌 Lc. garvieae 意大利黑麦草 2 ED-IR 耐久肠球菌 E. durans 意大利黑麦草 10 LcL-SC 乳酸乳球菌 Lc. lactis 甜玉米 3 EG-IR 灰黄色肠球菌 E. gilvus 意大利黑麦草 11 LeC-IR 柠檬明串珠菌 Le. citreum 意大利黑麦草 4 LbB-KG 短乳杆菌 Lb. brevis 热研四号王草 12 LeM-IR 肠膜明串珠菌 Le. mesenteroides 意大利黑麦草 5 LbF-WM 发酵乳杆菌 Lb. fermentum 糯玉米 13 LeP-IR 假肠膜明串珠菌 Le. pseudomesenteroides 意大利黑麦草 6 LbPe-EG 戊糖乳杆菌 Lb. pentosus 摩特矮象草 14 WCo-KG 融合乳杆菌 W. confusa 热研四号王草 7 LbPl-IR 植物乳杆菌 Lb. plantarum 意大利黑麦草 15 WCi-MT 魏斯特菌 W. cibaria MT-1杂交象草 8 LbR-EG 罗伊氏乳杆菌 Lb. reuteri 摩特矮象草 16 WP-IR 类肠膜魏斯氏菌 W. paramesenteroides 意大利黑麦草 表 2 牧草原料中糖分组成
Table 2. Sugar composition in forage raw materials (%)
项目
Item葡萄糖
Glucose蔗糖
Sucrose果糖
Fructose棉籽糖
Raffinose木糖
Xylose热研四号王草
Pennisetum purpureum × P. americanum cv. Reyan No.40.07±0.01c 1.91±0.37a 1.34±0.23b 1.86±0.06a 0.00±0.00c MT-1象草
P. purpureum cv. MT-10.10±0.01c 1.82±0.08a 1.41±0.25b 1.28±0.27b 0.03±0.00c 摩特矮象草
P. purputeum cv. Mott0.12±0.01c 1.75±0.29ab 2.11±0.13a 1.58±0.31b 0.09±0.09c 甜玉米
Zea mays L. saccharata Sturt.1.84±0.26b 6.00±0.96a 1.12±0.11b 0.00±0.00c 0.00±0.00c 桂牧一号杂交象草
(P. americanum × P. Purpureum) × (P. Purpureum cv. Mott) Guimu No.10.03±0.02d 1.87±0.14a 0.21±0.07c 1.24±0.17b 0.00±0.00d 糯玉米
Zea mays L. Sinensisa Kulesh1.81±0.36b 5.06±0.37a 0.93±0.02c 0.00±0.00d 0.00±0.00d 意大利黑麦草
Lolium multiflorum0.70±0.39ab 0.94±0.36a 0.34±0.07b 0.08±0.08c 0.10±0.04c 同行数据后不同小写字母表示差异显著(P<0.05),相同或无字母表示差异不显著(P>0.05);下表同。
Values in the same row with different small letter superscripts means significant difference (P<0.05), while with the same or no letter superscripts mean so significant difference (P>0.05). The same as below.表 3 16株乳酸菌对6种糖源培养液发酵后的碳源利用率
Table 3. The utilization rate of 16 strains of LAB to 6 kinds of sugar treatments (%)
菌株编号
Strain code葡萄糖MRS培养基
MRS-P果糖MRS培养基
MRS-G棉籽糖MRS培养基
MRS-MZ蔗糖MRS培养基
MRS-Z木糖MRS培养基
MRS-M混合糖MRS培养基
MRS- MIXEF-KG 94.68±1.01a 29.25±3.43e 0.73±0.53f 89.99±1.72b 59.95±1.48c 50.27±0.54d ED-IR 44.76±2.10b 60.48±1.52a 63.67±0.91a 69.87±1.16a 5.22±0.18c 39.83±2.34b EG-IR 71.44±3.35ab 58.97±3.27bc 49.20±1.22cd 81.23±1.54a 0.00±0.00e 39.56±0.89d LbB-KG 72.13±1.05a 18.10±0.53c 7.80±4.49c 50.41±0.10b 46.74±0.10b 38.82±2.37b LbF-WM 87.50±1.43b 100.00±0.00a 100.00±0.00a 89.79±3.33b 79.15±0.84b 33.33±4.13c LbPe-EG 82.97±0.73a 60.31±0.62b 37.42±3.02c 76.75±1.05ab 1.10±0.19d 38.56±2.88c LbPl-IR 69.56±1.06a 58.45±1.72a 60.34±1.81a 72.80±2.56a 0c 39.57±2.86b LbR-EG 82.07±2.63b 100.00±0.00a 100.00±0.00a 100.00±0.00a 0d 31.01±5.99c LcG-IR 78.09±0.84ab 60.59±1.02b 74.69±0.71ab 81.48±1.64a 1.92±0.42d 38.11±3.78c LcL-SC 93.58±1.01a 0e 18.52±0.52d 90.29±0.82a 37.25±0.12c 52.81±5.62b LeC-IR 83.17±3.13b 79.15±0.61b 10.49±0.03d 100.00±0.00a 15.54±0.16d 44.51±10.98c LeM-IR 73.89±1.05a 55.36±4.22b 70.94±0.01a 74.02±0.65a 0d 24.70±4.80c LeP-IR 80.19±1.64b 90.59±0.10a 100.00±0.00a 52.23±6.10c 24.86±20.14c 33.81±2.39c WCo-KG 76.66±1.64b 9.93±5.08e 17.11±1.42d 96.40±1.00a 48.27±0.10c 53.27±6.55c WCi-MT 67.93±3.46a 56.89±0.82a 68.83±1.55a 71.00±0.86a 8.80±0.18b 27.70±7.52b WP-IR 79.14±1.24a 73.84±0.01a 79.16±2.21a 79.09±1.13a 0c 41.12±7.76b 表 4 16株乳酸菌在不同碳源培养基发酵中的乳酸产量
Table 4. The production of lactic acid by 16 strains of LAB in different mediums (%)
菌株名称
Name葡萄糖MRS培养基
MRS-P果糖MRS培养基
MRS-G棉籽糖MRS培养基
MRS-MZ蔗糖MRS培养基
MRS-Z木糖MRS培养基
MRS-M混合糖MRS培养基
MRS- MIXEF-KG 0.69±0.01a 0.32±0.02b 0. 01±0.00c 0.62±0.01a 0.58±0.03a 0.55±0.02a ED-IR 0.89±0.02a 0.85±0.05a 0.21±0.03b 1.14±0.01a 0.02±0.00c 0.86±0.06a EG-IR 1.25±0.05a 0.73±0.05b 0.66±0.03b 0.87±0.00b 0.01±0.00c 1.00±0.03a LbB-KG 0.71±0.00a 0.29±0.03c 0.23±0.00c 0.56±0.06b 0.63±0.04b 0.61±0.04b LbF-WM 0.67±0.01b 0.36±0.04c 0.71±0.01b 0.73±0.00b 0.94±0.00a 0.67±0.05b LbPe-EG 1.08±0.04b 1.10±0.06a 0.43±0.04c 1.31±0.00a 0.01±0.00c 0.98±0.02b LbPl-IR 1.14±0.01a 1.07±0.05a 1.01±0.02a 1.23±0.03a 0.61±0.02b 0.77±0.00b LbR-EG 0.73±0.02ab 0.69±0.04ab 0.86±0.02a 0.55±0.03b 0.63±0.04ab 0.67±0.00 ab LcG-IR 1.18±0.04a 0.88±0.00b 1.02±0.04a 1.15±0.01b a 0.31±0.00c 1.08±0.05a LcL-SC 0.69±0.02a 0.45±0.06b 0.15±0.00c 0.52±0.05ab 0.50±0.02ab 0.58±0.00a LeC-IR 0.71±0.02a 0.67±0.02a 0.14±0.05c 0.42±0.02b 0.15±0.00c 0.45±0.01b LeM-IR 1.13±0.04a 1.01±0.05a 1.16±0.00a 1.13±0.00a 0.71±0.01b 1.06±0.02a LeP-IR 0.79±0.00a 0.54±0.02c 0.76±0.02a 0.65±0.01bc 0.78±0.00a 0.61±0.01bc WCo-KG 0.60±0.02a 0.43±0.00b 0.13±0.00c 0.62±0.06a 0.55±0.02a 0.50±0.02ab WCi-MT 1.11±0.05a 0.54±0.00b 0.36±0.02c 0.67±0.05b 0.10±0.01d 0.69±0.02b WP-IR 0.58±0.00a 0.52±0.00a 0.08±0.01c 0.32±0.00b 0.64±0.02a 0.31±0.00b 表 5 16株乳酸菌的产酸效率
Table 5. Acid production efficiency of 16 strains of LAB (%)
菌株名称
Name葡萄糖MRS培养基
MRS-P果糖MRS培养基
MRS-G棉籽糖MRS培养基
MRS-MZ蔗糖MRS培养基
MRS-Z木糖MRS培养基
MRS-M混合糖MRS培养基
MRS- MIXEF-KG 36.44±0.01c 54.69±0.06a 34.26±1.17d 34.45±0.01d 48.38±0.06b 54.71±0.59a ED-IR 99.41±0.22a 70.27±0.02d 16.49±0.00f 81.58±0.07b 19.16±0.66e 71.47±0.00c EG-IR 87.49±0.06a 61.90±0.02d 67.07±0.02c 53.55±0.03e 50.00±0.00f 82.72±0.00b LbB-KG 49.21±0.04b 80.12±0.15ab 81.69±0.20a 55.55±0.11b 67.40±0.15ab 49.85±0.00b LbF-WM 38.29±0.01d 18.00±0.00f 35.50±0.00e 40.65±0.01c 59.38±0.03a 49.12±0.00b LbPe-EG 65.08±0.02d 91.20±0.03a 57.46±0.03e 85.34±0.06b 23.10±3.98f 79.76±0.00c LbPl-IR 81.94±0.06d 91.53±0.03a 83.69±0.01c 84.70±0.45b 81.33±0.00e 63.71±0.00f LbR-EG 44.48±0.02c 34.50±0.00e 43.00±0.00d 27.50±0.00f 45.99±0.00b 48.56±0.00a LcG-IR 75.55±0.04b 72.61±0.02c 68.28±0.01e 70.57±0.03d 41.77±0.00f 87.25±0.00a LcL-SC 36.87±0.01d 66.18±0.00b 40.49±0.05e 28.80±0.01f 67.11±0.22a 61.45±0.00c LeC-IR 42.69±0.02c 42.32±0.01c 66.75±0.14a 21.00±0.00e 48.28±0.50b 40.55±0.00d LeM-IR 76.47±0.05d 91.22±0.03a 81.76±0.01b 76.33±0.05e 79.78±0.00c 70.38±0.00f LeP-IR 49.26±0.03c 29.80±0.01f 38.00±0.00e 62.23±0.12b 86.66±0.00a 46.08±0.00d WCo-KG 39.13±0.02d 43.10±0.00c 37.98±0.05e 32.16±0.00f 56.97±0.12a 53.50±0.00b WCi-MT 81.70±0.07a 47.46±0.02c 26.15±0.00d 47.18±0.04c 56.85±1.12b 47.72±0.00c WP-IR 36.65±0.02a 35.21±0.01a 5.05±0.00b 20.23±0.01ab 15.80±27.37ab 26.33±0.00ab -
[1] 张志飞, 王青兰. 牧草青贮乳酸菌研究进展 [J]. 湖南生态科学学报, 2021, 8(1):70−76. doi: 10.3969/j.issn.2095-7300.2021.01.010ZHANG Z F, WANG Q L. Research progress of lactic acid bacteria in forage silage [J]. Journal of Hunan Ecological Science, 2021, 8(1): 70−76. (in Chinese) doi: 10.3969/j.issn.2095-7300.2021.01.010 [2] 杨俊峰, 沈向华, 金曙光. 乳酸菌在饲料青贮中的应用及研究进展 [J]. 浙江畜牧兽医, 2011, 36(5):9−11. doi: 10.3969/j.issn.1005-7307.2011.05.005YANG J F, SHEN X H, JIN S G. Application and research progress of lactic acid bacteria in feed silage [J]. Zhejiang Journal Animal Science and Veterinary Medicine, 2011, 36(5): 9−11. (in Chinese) doi: 10.3969/j.issn.1005-7307.2011.05.005 [3] 李新一, 程晨, 尹晓飞, 等. 中外草牧业发展历程、重点与中国草牧业发展措施 [J]. 草原与草业, 2020, 32(4):6−13. doi: 10.3969/j.issn.2095-5952.2020.04.007LI X Y, CHENG C, YIN X F, et al. Comparative analysis and enlightening countermeasures of Chinese and foreign grass and husbandry development [J]. Grassland and Prataculture, 2020, 32(4): 6−13. (in Chinese) doi: 10.3969/j.issn.2095-5952.2020.04.007 [4] 杨楠, 邹苏燕, 戚如鑫, 等. 青贮微生物制剂及优良青贮菌种筛选的研究进展 [J]. 动物营养学报, 2020, 32(2):578−585. doi: 10.3969/j.issn.1006-267x.2020.02.013YANG N, ZOU S Y, QI R X, et al. Research progress on microbial preparations of silage and selection of optimum strains for silage preparing [J]. Chinese Journal of Animal Nutrition, 2020, 32(2): 578−585. (in Chinese) doi: 10.3969/j.issn.1006-267x.2020.02.013 [5] 吕竑建, 郭香, 陈德奎, 等. 植物乳酸菌和贮藏温度对辣木叶青贮品质的影响 [J]. 草业学报, 2021, 30(3):121−128. doi: 10.11686/cyxb2020152LV H J, GUO X, CHEN D K, et al. Effect of lactic acid bacteria and storage temperature on the quality of Moringa oleifera leaf silage [J]. Acta Prataculturae Sinica, 2021, 30(3): 121−128. (in Chinese) doi: 10.11686/cyxb2020152 [6] CAI Y M, BENNO Y, OGAWA M, et al. Influence of Lactobacillus spp. from an inoculant and of Weissella and Leuconostoc spp. from forage crops on silage fermentation [J]. Applied and Environmental Microbiology, 1998, 64(8): 2982−2987. doi: 10.1128/AEM.64.8.2982-2987.1998 [7] 蔡红英. 植物乳杆菌对小鼠肝脏脂代谢的调控与作用机制[D]. 北京: 中国农业科学院, 2020.CAI H Y. Effects and Mechanisms of Hepatic Lipid Regulation in Mice by Lactobacillus Plantarum[D]. Beijing: Chinese Academy of Agricultural Sciences, 2020. (in Chinese) [8] LINDOW S E, BRANDL M T. Microbiology of the phyllosphere [J]. Applied and Environmental Microbiology, 2003, 69(4): 1875−1883. doi: 10.1128/AEM.69.4.1875-1883.2003 [9] MERCIER J, LINDOW S E. Role of leaf surface sugars in colonization of plants by bacterial epiphytes [J]. Applied and Environmental Microbiology, 2000, 66(1): 369−374. doi: 10.1128/AEM.66.1.369-374.2000 [10] 郝永伟, 吕蕾. 植物乳杆菌增菌培养基碳源优化 [J]. 齐鲁工业大学学报, 2014, 28(1):36−38.HAO Y W, LV L. Optimization of carbon of culture medium for accumulating Lactobacillus plantarum [J]. Journal of Qilu University of Technology, 2014, 28(1): 36−38. (in Chinese) [11] 金玉洁, 何国庆. 植物乳杆菌ZU018增殖培养基的优化[J]. 食品工业科技, 2020, 41(14): 94, 100, 95-99.JIN Y J, HE G Q. Optimization of Lactobacillus plantarum ZU018 proliferation medium[J]. Science and Technology of Food Industry, 2020, 41(14): 94, 95-99, 100. (in Chinese) [12] 洪梅, 刁其玉, 闫贵龙, 等. 响应面法优化青贮饲料乳酸菌的培养条件 [J]. 动物营养学报, 2010, 22(5):1307−1313. doi: 10.3969/j.issn.1006-267x.2010.05.027HONG M, DIAO Q Y, YAN G L, et al. Optimization of culture conditions for silage lactic acid bacteria via a response surface technique [J]. Chinese Journal of Animal Nutrition, 2010, 22(5): 1307−1313. (in Chinese) doi: 10.3969/j.issn.1006-267x.2010.05.027 [13] CHEN X Z, ZHUANG Y F, DONG Z X, et al. Factors influencing the distribution of lactic acid bacteria on Pennisetum grasses [J]. Grassland Science, 2017, 63(3): 150−158. doi: 10.1111/grs.12161 [14] 范佳硕, 杨鑫, 陈博, 等. 植物乳杆菌发酵培养基优化及工艺开发 [J]. 发酵科技通讯, 2020, 49(1):21−26.FAN J S, YANG X, CHEN B, et al. Optimization of Lactobacillus plantarum fermentation medium and process development [J]. Bulletin of Fermentation Science and Technology, 2020, 49(1): 21−26. (in Chinese) [15] ZHANG J, TANAKA O, UEGAKI R, et al. The effect of inoculation and additives on D (–) - and L(+)-lactic acid production and fermentation quality of guineagrass (Panicum maximum Jacq) silage [J]. Journal of the Science of Food and Agriculture, 2000, 80(15): 2186−2189. doi: 10.1002/1097-0010(200012)80:15<2186::AID-JSFA767>3.0.CO;2-1 [16] 陈绮, 雷文平, 肖茜, 等. 植物乳杆菌XN1904E产胞外多糖发酵条件优化 [J]. 乳业科学与技术, 2020, 43(3):1−5.CHEN Q, LEI W P, XIAO Q, et al. Optimization of fermentation conditions for exopolysaccharide production by Lactobacillus plantarum XN1904E [J]. Journal of Dairy Science and Technology, 2020, 43(3): 1−5. (in Chinese) [17] 陶静, 许赛信, 孟德俊, 等. 乳酸菌发酵生产胞外多糖条件优化研究 [J]. 食品工业, 2017, 38(1):28−31.TAO J, XU S X, MENG D J, et al. The conditions optimization of Lactobacillus exopolysaccharides fermentation [J]. The Food Industry, 2017, 38(1): 28−31. (in Chinese) [18] 刘金玲, 李嘉文, 张含雪, 等. 罗伊乳杆菌增殖培养基中碳源氮源的优化 [J]. 中国微生态学杂志, 2016, 28(5):533−537.LIU J L, LI J W, ZHANG H X, et al. Optimization of carbon and nitrogen sources of enrichment culture medium for Lactobacillus reuteri [J]. Chinese Journal of Microecology, 2016, 28(5): 533−537. (in Chinese) [19] 季海蕊, 郭尚旭, 姜静, 等. 乳酸明串珠菌(Leuconostoc lactis)L2体内耐受性及产胞外多糖条件研究 [J]. 黑龙江大学自然科学学报, 2020, 37(5):580−587.JI H R, GUO S X, JIANG J, et al. The in vivo tolerance of Leuconostoc lactis L2 and its condition study on exopolysaccharide production [J]. Journal of Natural Science of Heilongjiang University, 2020, 37(5): 580−587. (in Chinese) [20] 李想, 王然, 程建军, 等. 植物乳杆菌培养基的优化 [J]. 东北农业大学学报, 2008, 39(9):96−99. doi: 10.3969/j.issn.1005-9369.2008.09.023LI X, WANG R, CHENG J J, et al. Optimization of culture medium for L. plantarum [J]. Journal of Northeast Agricultural University, 2008, 39(9): 96−99. (in Chinese) doi: 10.3969/j.issn.1005-9369.2008.09.023 [21] 岳苗苗, 汪岚, 唐国建, 等. 青贮用乳酸菌的高效保存方法研究 [J]. 饲料研究, 2020, 43(10):73−77.YUE M M, WANG L, TANG G J, et al. Research on efficient preservation of lactic acid bacteria as silage additive [J]. Feed Research, 2020, 43(10): 73−77. (in Chinese) [22] 刘毓锋, 曾嘉锐, 黄文琪, 等. 外源碳源对葡萄酵素微生物生长代谢及生物活性的调节作用 [J]. 食品工业科技, 2020, 41(8):104−110,116.LIU Y F, ZENG J R, HUANG W Q, et al. Regulating effect of exogenous carbon source on microbial growth and metabolism in grape fermentation and its bioactivity [J]. Science and Technology of Food Industry, 2020, 41(8): 104−110,116. (in Chinese) [23] 程方方, 杨连玉, 许雪魁, 等. 糖蜜和乳酸菌对光叶紫花苕青贮品质的影响 [J]. 草地学报, 2012, 20(5):947−951.CHENG F F, YANG L Y, XU X K, et al. Effect of adding molasses and lactic acid bacteria on the silage quality of smooth vetch [J]. Acta Agrestia Sinica, 2012, 20(5): 947−951. (in Chinese) [24] 荣辉, 余成群, 李志华, 等. 添加糖蜜和尿素对象草青贮发酵品质的影响 [J]. 草地学报, 2012, 20(5):940−946.RONG H, YU C Q, LI Z H, et al. Effects of adding molasses and urea on fermentation quality of Napier grass silage [J]. Acta Agrestia Sinica, 2012, 20(5): 940−946. (in Chinese) [25] YOKOTA H, KIM J H, OKAJIMA T, et al. Nutritional quality of wilted Napier grass (Pennisetum purpureum Schum. ) ensiled with or without molasses [J]. Asian-Australasian Journal of Animal Sciences, 1992, 5(4): 673−679. doi: 10.5713/ajas.1992.673 [26] 郭金双, 赵广永, 杨雅芳, 等. 添加蔗糖对大麦青贮品质及中、酸性洗涤纤维瘤胃降解率的影响 [J]. 中国畜牧杂志, 2000, 36(4):18−20. doi: 10.3969/j.issn.0258-7033.2000.04.007GUO J S, ZHAO G Y, YANG Y F, et al. Effect of sugar supplementation on the quality of barley silage and NDF and ADF degradation in the rumen of cattle [J]. Chinese Journal of Animal Science, 2000, 36(4): 18−20. (in Chinese) doi: 10.3969/j.issn.0258-7033.2000.04.007 [27] 万里强, 李向林. 乳酸菌复合添加剂糖分浓度对不同含水量苜蓿青贮效果的影响 [J]. 中国草地, 2005, 27(1):45−51.WAN L Q, LI X L. Influence of sugar contents in lactic bacteria compound additives on different moisture alfalfa silage [J]. Grassland of China, 2005, 27(1): 45−51. (in Chinese) -






 下载:
下载:

