Cloning and Expression of Premnaspirodiene Oxygenase Gene from Dendrobium officinale
-
摘要:目的 克隆铁皮石斛(Dendrobium officinale)豆腐柴螺二烯加氧酶(premnaspirodiene oxygenase, PO)基因,分析该基因在铁皮石斛开花期不同器官及营养生长期叶片中的表达模式,以期为进一步鉴定基因功能及探讨铁皮石斛植保素的生物合成及抗病机制奠定基础。方法 利用RT-PCR和RACE-PCR技术克隆DoPO基因全长cDNA序列和开放阅读框(open reading frame, ORF),利用ProtParam进行理化性质分析,使用BLAST P在线搜索氨基酸同源性,采用MEGA 6.0构建系统进化树。利用qPCR方法分析DoPO基因在铁皮石斛开花期不同器官及不同营养生长期叶片中的表达模式。结果 DoPO基因cDNA序列全长1704 bp,含1个编码498个氨基酸的完整阅读框。DoPO蛋白属于p450超级家族,分子量为56.914 kD,属不稳定蛋白。系统进化分析表明,DoPO与兰科植物姬蝴蝶兰(XP 020571355)和深圳拟兰(PKA52400)PO聚为一类,与姬蝴蝶兰的PO亲缘关系最近。qPCR分析结果表明,铁皮石斛开花期,DoPO基因在叶片的相对表达量极显著高于茎、根和花;叶片DoPO基因的相对表达量10月份极显著高于8月和12月,12月显著高于8月。结论 克隆了铁皮石斛DoPO基因cDNA全长,该基因在叶片中的相对表达量极显著高于其他器官。叶片DoPO基因的相对表达量10月份最高。Abstract:Objective To decipher the gene function, phytoalexin biosynthesis, and disease resistance mechanism of Dendrobium officinale (Do), the premnaspirodiene oxygenase
gene (PO) was cloned and its expressions in the plant in various organs at different growth stages studied. Method RACE-PCR and RT-PCR were employed to clone the full-length cDNA and ORF of DoPO. ProtParam was used for the physiochemical determination, BLAST P on the amino acid homology, and MEGA 6.0 for the phylogenetic tree construction. The expressions of DoPO in different organs at flowering stage as well as in leaves at growth stages were examined by qPCR.Result The DoPO encoded 498 amino acids, 1 704 bp in length with an 1 497 bp ORF. The unstable protein had a molecular weight of 56.914 kDa belonging to the P450 superfamily. It was phylogenetically clustered in the same branch with Phalaenopsis equestris (XP 020571355) and Apostasia shenzhenica (PKA52400) and most closely related to P. equestris. The relative DoPO expression in the leaves was significantly higher than that in the stems, roots, or flowers at flowering stage (P<0.01); and during a year, that in October significantly higher than that in either August or December (P<0.01) and that in December significantly higher than in August (P<0.05).Conclusion The full-length cDNA of DoPO was successfully cloned. Its relative expression in leaf peaked in October and was highest in leaves among the organs at flowering stage. -
0. 引言
【研究意义】植保素(Phytoalexin,又称植物抗毒素)是一类具有物种特异性的小分子有机化合物,具有抗菌和驱虫活性,受到病原菌或害虫诱导产生[1]。目前已经鉴定的植保素主要有黄酮类、萜类、芪类、吲哚类、生物碱类和香豆素类等[2-3]。禾本科(Poaceae)植物主要产生二萜或黄酮类植保素[4-5]、豆科(Fabaceae)植物主要产生异黄酮类植保素[6]、茄科(Solanaceae)植物主要产生萜类植保素[7-8]。螺岩兰草酮(Solavetivone)是一种倍半萜类植保素。豆腐柴螺二烯(Premnaspirodiene)合成酶催化法呢基二磷酸(FPP)环化形成豆腐柴螺二烯[9],豆腐柴螺二烯在豆腐柴螺二烯加氧酶(Premnaspirodiene oxygenase)催化下,经过羟基化和氧化形成[10]螺岩兰草酮。因此,豆腐柴螺二烯加氧酶是螺岩兰草酮合成的关键酶。【前人研究进展】黄果茄(Solanum xanthocarpum)是一种重要的民间医药,具有杀虫、抗菌[11-12]等功效,其根系提取的精油中,螺岩兰草酮占22.9%[13]。DESJARDINS等[14]研究发现,马铃薯(Solanum tuberosum)受到线虫侵染后,抗病品种和感病品种根系的倍半萜烯含量变化不大,但是螺岩兰草酮在倍半萜烯中的比例显著提高。YAO等[15]研究结果表明马铃薯试管苗用丛枝菌根菌(Glomus etunicatum)菌根化后受到立枯丝核菌(Rhizoctonia solani)侵染,根部螺岩兰草酮和日齐素(Rishitin)含量显著增加,通过培养皿生物测定,螺岩兰草酮和日齐素抑制立枯丝核菌菌丝生长。KAWAUCHI[16-17]等用茉莉酸甲酯和硫酸铜处理天仙子(Hyoscyamus albus)毛状根,会诱导合成螺岩兰草酮及其衍生物,并克隆了螺岩兰草酮合成酶基因豆腐柴螺二烯加氧酶基因(hahpo1)。【本研究切入点】铁皮石斛(Dendrobium officinale)属兰科(Orchidaceae)石斛属(Dendrobium Sw.)药用植物,具有增强免疫力、抑制肿瘤等功效[18-21]。目前,鲜见铁皮石斛豆腐柴螺二烯加氧酶基因的相关研究报道。【拟解决的关键问题】本课题组从不同营养生长期铁皮石斛叶片转录组测序数据中筛选差异表达基因时,获得1条带5′末端的铁皮石斛豆腐柴螺二烯加氧酶基因cDNA片段,根据cDNA序列信息设计3′RACE引物,利用RT-PCR技术克隆基因全长cDNA序列和ORF,分析豆腐柴螺二烯加氧酶基因在铁皮石斛不同营养生长阶段和开花期不同器官中的表达,以期为进一步鉴定基因功能及探讨铁皮石斛植保素的生物合成及抗病机制奠定基础。
1. 材料与方法
1.1 试验材料
1.1.1 植物材料
冠豸山铁皮石斛开花期的根、茎、叶和花及8月、10月和12月的叶片。
1.1.2 试验试剂
宝生物工程有限公司的PrimeScriptTM Reverse Transcriptase和pMD19-T;湖南艾科瑞生物工程有限公司的SteadyPure 植物RNA提取试剂盒、2×Accurate Taq Master Mix(dye plus)和SYBR Green Pro Taq HS 预混型qPCR试剂盒;测序委托生工生物工程(上海)股份有限公司完成。
1.2 DoPO基因克隆
根据转录组测序获得的带5′端的DoPO基因序列信息设计3′RACE、克隆开放阅读框引物及3′RACE接头引物,引物见表1,下同。由冠豸山铁皮石斛叶片提取的总RNA逆转录的单链cDNA为模板进行PCR扩增,PCR产物经克隆后测序。
表 1 PCR引物及其序列Table 1. PCR primers and sequences引物名称
Primer name碱基序列(5'-3')
Primer sequence(5’-3')3PO-1 GCTGGAACTGACACCTCATCTG 3PO-2 CCAGCAGGAAGTCGGATTGTGA Adpt CTGATCTAGAGGTACCGGATCC dT-Adpt CTGATCTAGAG dGTACCGGATCC
TTTTTTTTTTTTTTTTTDoPO-F CCGGGATCCATGGAGCCACCAACCTTC DoPO-R CCGCTCGAGACGTAGGCAATGGAGGAT PO-F GCTCTTGTCGGGATTCAG PO-R GATTCTTTGCTCGATGCTC DoACT-F AGGAAGGCGGCTTTGAATC DoACT-R CCATGCCAACCATGACACC 注:下划线碱基为酶切位点。
Note: The underlined bases were the enzyme site.1.3 生物信息学分析
利用BLAST P在GeneBank中搜索DoPO氨基酸序列相似性和保守结构域。利用ProtParam预测DoPO蛋白的理化性质。系统进化分析(Neighbor Joining Tree, Bootstrap 1000)和进化树构建由MEGA 6.0软件完成[22]。
1.4 DoPO基因表达分析
按照SteadyPure 植物RNA提取试剂盒的说明提取各样品的RNA,用PrimeScriptTM Reverse Transcriptase逆转录成cDNA,浓度定量为200 ng·μL−1。设计qPCR引物PO-F和PO-R,扩增长度为149 bp。Actin基因属于持家基因,在细胞中组成型表达,常用于基因表达分析的内参基因。选择铁皮石斛Actin作为内参基因,引物为DoACT-F和DoACT-R,产物长度为215 bp。实时荧光定量PCR参考林江波等的方法[23]。
2. 结果与分析
2.1 DoPO基因的3′RACE克隆
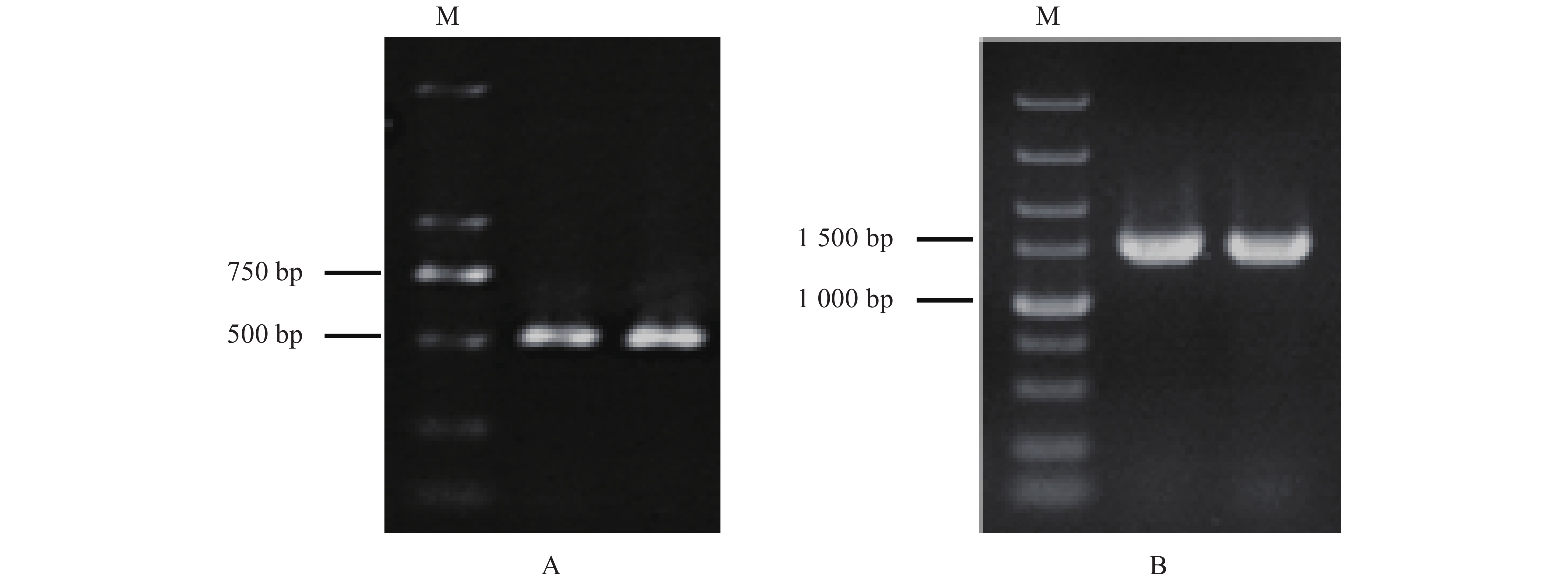
用随机引物反转录的cDNA为模板,用引物3PO-1和dT-Adpt、引物3PO-2和Adpt先后进行二轮PCR,电泳结果见图1A,在500 bp左右有1条特异性条带。胶回收目的片段,经过克隆和测序后,用DNAMAN V6.0对3′-RACE的测序结果进行序列拼接和ORF的分析。结果表明获得DoPO基因cDNA全长,长度为1704 bp (图2),含1个编码498个氨基酸的完整阅读框架,5′末端非翻译区40 bp,3′末端非翻译区167 bp。
2.2 DoPO基因ORF克隆
以随机引物反转录的铁皮石斛叶片cDNA为模板,利用引物DoPO-F和DoPO-R进行PCR扩增,PCR产物电泳结果(图1B)显示,在1532 bp处有1条特异性条带。PCR产物经过克隆和测序,结果表明:获得了带BamH I和Xho I酶切位点的DoPO基因ORF。提取质粒pMD19-T-DoPO,-20℃保存。
2.3 生物信息学分析
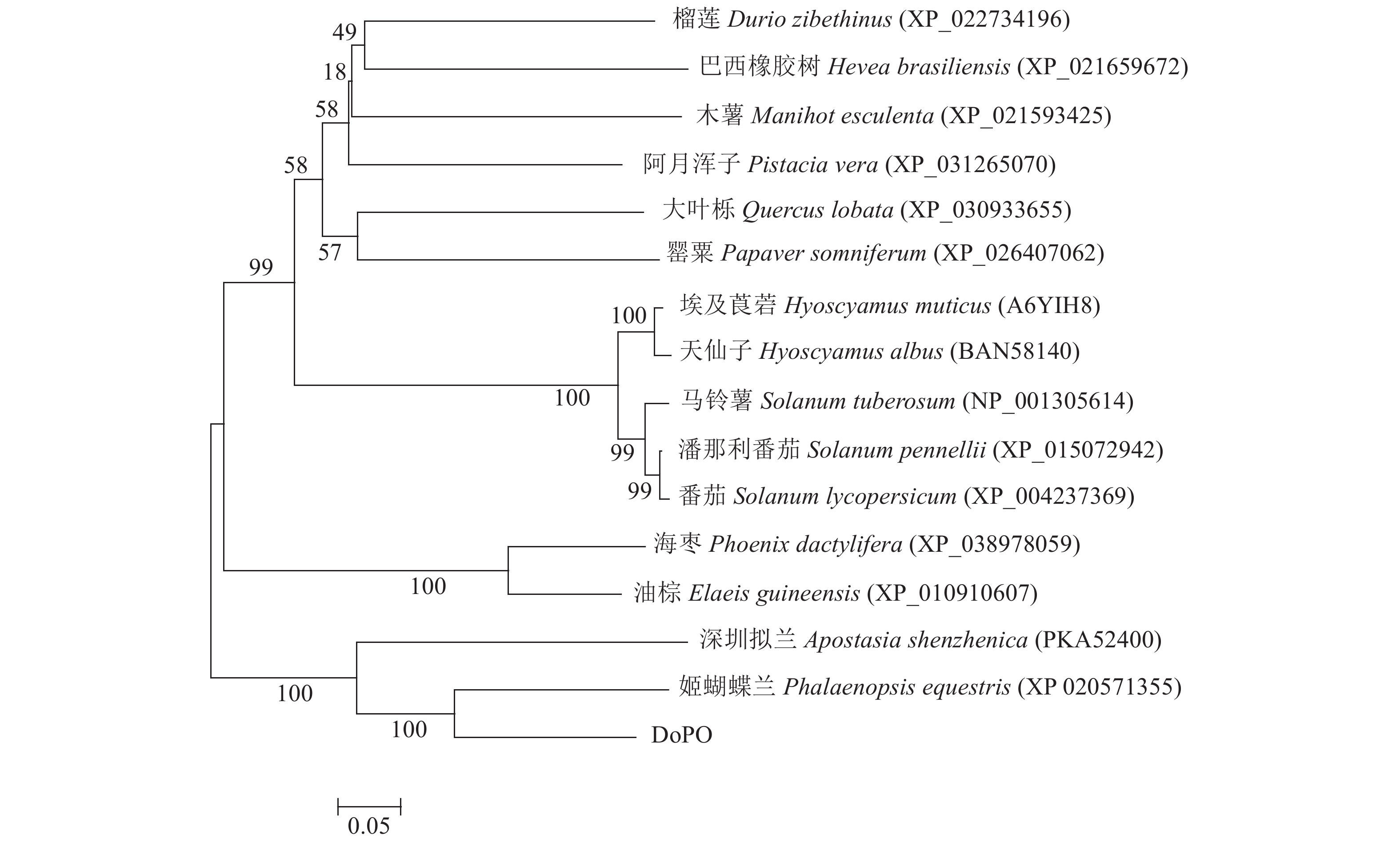
DoPO蛋白的分子量为56.914 kD,属不稳定蛋白。使用BLAST P检索DoPO蛋白保守结构域和同源序列,结果表明:DoPO蛋白属于p450超级家族,氨基酸序列与姬蝴蝶兰(Phalaenopsis equestris, XP 020571355)、深圳拟兰(Apostasia shenzhenica, PKA52400)和埃及莨菪(Hyoscyamus muticus, A6YIH8)的PO氨基酸相似度分别为85%、76%和70%。将DoPO与其他15个物种的PO氨基酸序列进行系统进化分析,结果表明(图3):16个物种聚为4大类,铁皮石斛与兰科植物的姬蝴蝶兰(XP 020571355)和深圳拟兰(PKA52400)聚为一类,与姬蝴蝶兰亲缘关系最近;棕榈科的海枣(Phoenix dactylifera, XP_038978059)和油棕(Elaeis guineensis, XP_010910607)聚为一类;茄科植物的埃及莨菪(Hyoscyamus muticus, A6YIH8)、天仙子(Hyoscyamus albus, BAN58140)、马铃薯(Solanum tuberosum, NP_001305614)、潘那利番茄(Solanum pennellii, XP_015072942)和番茄(Solanum lycopersicum, XP_004237369)聚为一类。
2.4 DoPO基因在不同器官的表达
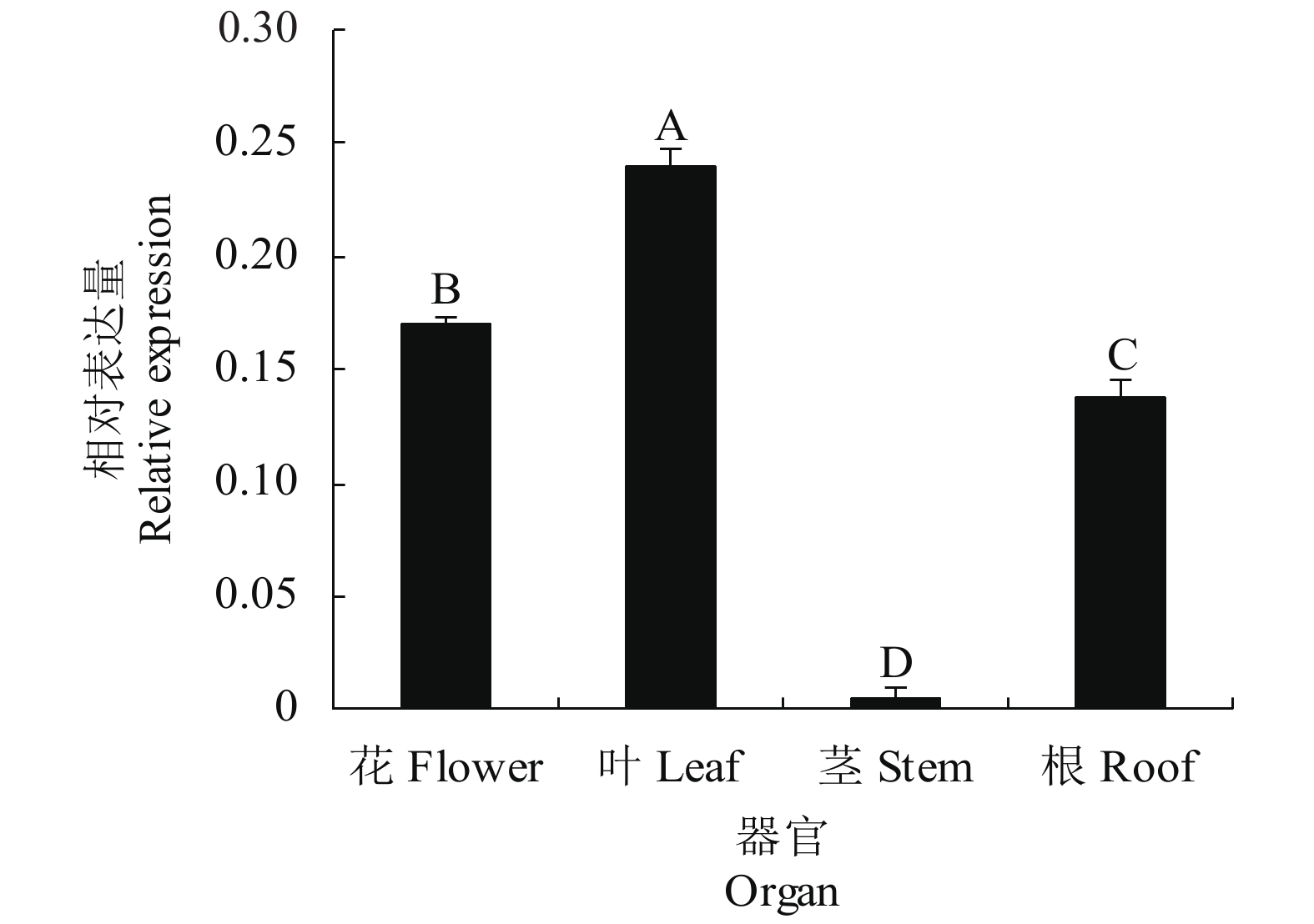
以Actin基因作为内参基因,qRT-PCR检测DoPO基因在铁皮石斛开花期不同器官的表达,结果表明(图4),花、茎、叶和根都能检测到DoPO基因的表达,表达量的高低依次为叶>花>根>茎,而且表达量存在极显著差异。
2.5 DoPO基因在不同营养生长期叶中的表达
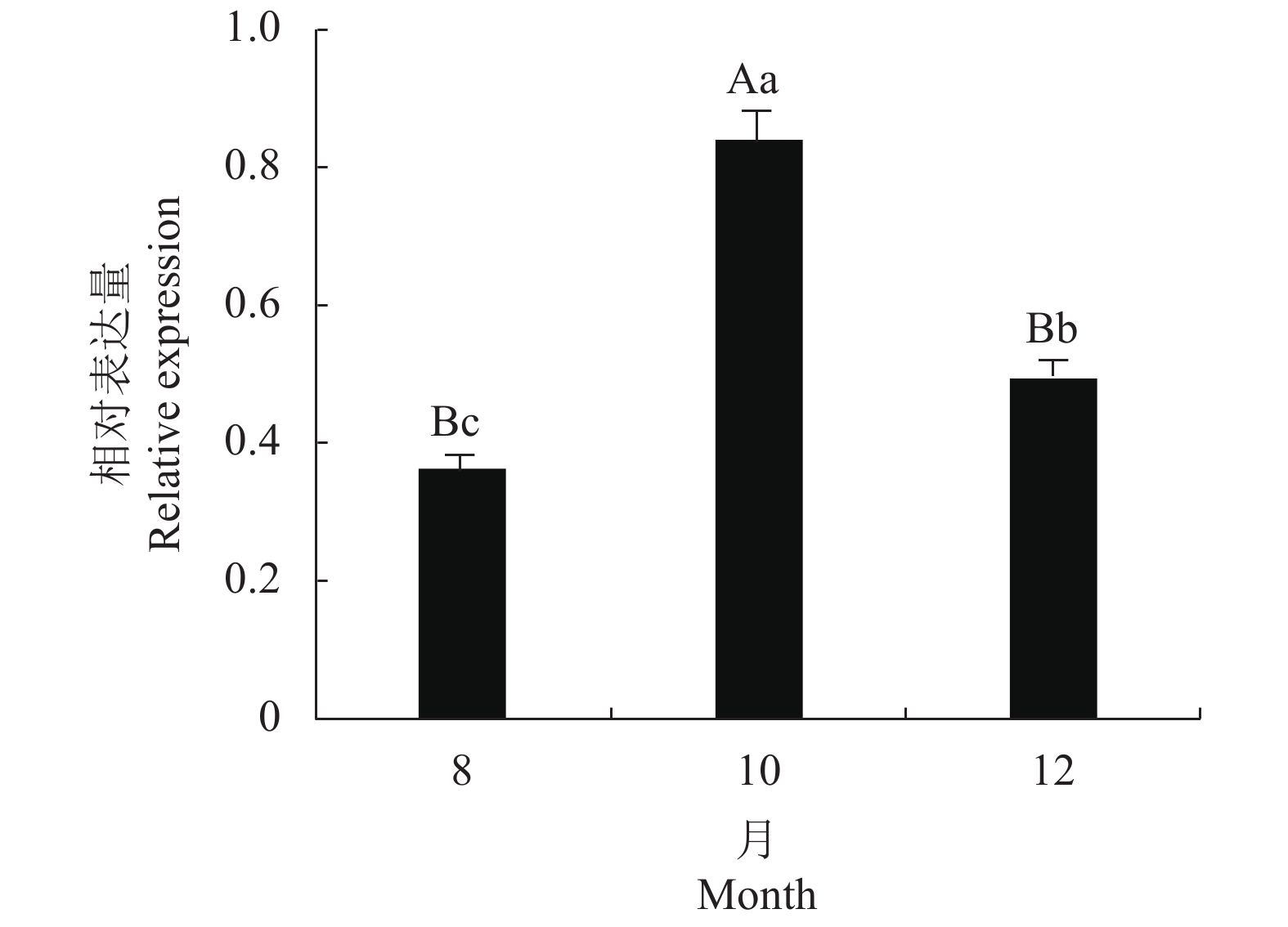
为了进一步分析DoPO基因在铁皮石斛不同营养生长阶段叶片中的表达模式,利用荧光定量PCR技术分析了铁皮石斛叶片DoPO基因在8、10、12月份的表达情况,结果表明(图5):10月份的表达量最高,与8、12月份的差异极显著。其次是12月份,与8月份的差异显著。
3. 讨论与结论
豆腐柴螺二烯加氧酶是植物合成植保素螺岩兰草酮的关键酶。TAKAHASHI等[10]克隆了埃及莨菪的HPO基因,其编码蛋白属于P450超级家族成员,体外酶活性实验表明,具有催化豆腐柴螺二烯形成螺岩兰草酮的活性;此外,还可以催化几种艾里莫芬烷型(十氢化萘环系统)倍半萜烯C-2的羟基化。细胞色素P450(cytochrome P450,CYP)酶是一类血红素蛋白类酶,具有氧化、羟基化和去甲基化等多种催化活性[24],如果氨基酸序列一致性高于40%属于同一个家族,高于55%则属于同一个亚家族[25]。本研究克隆了铁皮石斛的DoPO基因,属于P450超级家族,核苷酸序列与埃及莨菪HPO(A6YIH8)的氨基酸相似度为70%,属同一亚家族,推测DoPO具有催化豆腐柴螺二烯形成螺岩兰草酮的活性。
植保素一般在病原菌或害虫侵害后几个小时开始形成和累积[26],是植物对抗病原菌、害虫等侵害的一种防御机制[27-28]。KAWAUCHI等[16]克隆了天仙子的hahpo1基因,茉莉酸甲酯处理后24 h、茉莉酸甲酯和硫酸铜共同处理后6 h能检测到hahpo1基因的表达,未处理没有检测到表达。本研究发现铁皮石斛DoPO基因在叶、茎、根和花中都有表达,但叶的相对表达量最高,茎的相对表达量最低,叶片DoPO基因的相对表达量又以10月份最高。从铁皮石斛DoPO基因表达模式来看,在整个生长期,各器官可能都有合成一定量的螺岩兰草酮,这需要通过检测各器官的螺岩兰草酮含量来进一步验证。
本研究克隆了铁皮石斛DoPO基因,分析了其在不同器官和营养生长期叶片的表达模式,为进一步研究其催化功能和探讨铁皮石斛植保素的生物合成及抗病机制奠定基础。
-
表 1 PCR引物及其序列
Table 1 PCR primers and sequences
引物名称
Primer name碱基序列(5'-3')
Primer sequence(5’-3')3PO-1 GCTGGAACTGACACCTCATCTG 3PO-2 CCAGCAGGAAGTCGGATTGTGA Adpt CTGATCTAGAGGTACCGGATCC dT-Adpt CTGATCTAGAG dGTACCGGATCC
TTTTTTTTTTTTTTTTTDoPO-F CCGGGATCCATGGAGCCACCAACCTTC DoPO-R CCGCTCGAGACGTAGGCAATGGAGGAT PO-F GCTCTTGTCGGGATTCAG PO-R GATTCTTTGCTCGATGCTC DoACT-F AGGAAGGCGGCTTTGAATC DoACT-R CCATGCCAACCATGACACC 注:下划线碱基为酶切位点。
Note: The underlined bases were the enzyme site. -
[1] JEANDET P, CLÉMENT C, COUROT E, et al. Modulation of phytoalexin biosynthesis in engineered plants for disease resistance [J]. International Journal of Molecular Sciences, 2013, 14(7): 14136−14170. DOI: 10.3390/ijms140714136
[2] 陈东菊, 安敏敏, 李丽兰, 等. 植保素及其在增强作物抗性中的作用 [J]. 分子植物育种, 2017, 15(2):774−780. CHEN D J, AN M M, LI L L, et al. The mechanism of phytoalexin enhancing crop disease resistance [J]. Molecular Plant Breeding, 2017, 15(2): 774−780.(in Chinese)
[3] 吴劲松. 植物对病原微生物的“化学防御”: 植保素的生物合成及其分子调控机制 [J]. 应用生态学报, 2020, 31(7):2161−2167. WU J S. The “chemical defense” of plants against pathogenic microbes: Phytoalexins biosynthesis and molecular regulations [J]. Chinese Journal of Applied Ecology, 2020, 31(7): 2161−2167.(in Chinese)
[4] TOYOMASU T. Recent advances regarding diterpene cyclase genes in higher plants and fungi [J]. Bioscience, Biotechnology, and Biochemistry, 2008, 72(5): 1168−1175. DOI: 10.1271/bbb.80044
[5] POLONI A, SCHIRAWSKI J. Red card for pathogens: Phytoalexins in Sorghum and maize [J]. Molecules (Basel, Switzerland), 2014, 19(7): 9114−9133. DOI: 10.3390/molecules19079114
[6] CHEN K N, ERH M H, SU N W, et al. Soyfoods and soybean products: From traditional use to modern applications [J]. Applied Microbiology and Biotechnology, 2012, 96(1): 9−22. DOI: 10.1007/s00253-012-4330-7
[7] 李端, 周立刚, 王蓟花, 等. 茄科植保素的研究进展 [J]. 天然产物研究与开发, 2004, 16(1):84−87, 79. DOI: 10.3969/j.issn.1001-6880.2004.01.024 LI D, ZHOU L G, WANG J H, et al. Progress on phytoalexin research of solanaceous plants [J]. Natural Product Research and Development, 2004, 16(1): 84−87, 79.(in Chinese) DOI: 10.3969/j.issn.1001-6880.2004.01.024
[8] DESENDER S, ANDRIVON D, VAL F. Activation of defence reactions in Solanaceae: Where is the specificity? [J]. Cellular Microbiology, 2007, 9(1): 21−30. DOI: 10.1111/j.1462-5822.2006.00831.x
[9] BACK K, CHAPPELL J. Cloning and bacterial expression of a sesquiterpene cyclase from Hyoscyamus muticus and its molecular comparison to related terpene cyclases [J]. The Journal of Biological Chemistry, 1995, 270(13): 7375−7381. DOI: 10.1074/jbc.270.13.7375
[10] TAKAHASHI S, YEO Y S, ZHAO Y X, et al. Functional characterization of premnaspirodiene oxygenase, a cytochrome P450 catalyzing regio- and stereo-specific hydroxylations of diverse sesquiterpene substrates [J]. Journal of Biological Chemistry, 2007, 282(43): 31744−31754. DOI: 10.1074/jbc.M703378200
[11] MAHESH KUMAR P, MURUGAN K, KOVENDAN K, et al. Mosquito larvicidal and pupicidal efficacy of Solanum xanthocarpum (Family: Solanaceae) leaf extract and bacterial insecticide, Bacillus thuringiensis, against Culex quinquefasciatus Say (Diptera: Culicidae) [J]. Parasitology Research, 2012, 110(6): 2541−2550. DOI: 10.1007/s00436-011-2797-2
[12] PARDHI P, JAIN A P, GANESHPURKAR A, et al. Anti-microbial, Anti-oxidant and anthelmintic activity of crude extract of Solanum xanthocarpum [J]. Pharmacognosy Journal, 2010, 2(11): 400−404. DOI: 10.1016/S0975-3575(10)80022-7
[13] SATYAL P, MAHARJAN S, SETZER W N. Volatile constituents from the leaves, fruits (berries), stems and roots of Solanum xanthocarpum from Nepal [J]. Natural Product Communications, 2015, 10(2): 1934578X1501000.
[14] DESJARDINS A E, MCCORMICK S P, PLAISTED R L, et al. Association between solavetivone production and resistance to Globodera rostochiensis in potato [J]. Journal of Agricultural and Food Chemistry, 1997, 45(6): 2322−2326. DOI: 10.1021/jf960830p
[15] YAO M K, DÉSILETS H, CHARLES M T, et al. Effect of mycorrhization on the accumulation of rishitin and solavetivone in potato plantlets challenged with Rhizoctonia solani [J]. Mycorrhiza, 2003, 13(6): 333−336. DOI: 10.1007/s00572-003-0267-0
[16] KAWAUCHI M, ARIMA T, SHIROTA O, et al. Production of sesquiterpene-type phytoalexins by hairy roots of Hyoscyamus albus co-treated with cupper culfate and methyl jasmonate [J]. Chemical and Pharmaceutical Bulletin, 2010, 58(7): 934−938. DOI: 10.1248/cpb.58.934
[17] KAWAUCHI M, DE ARIMA T H, KUROYANAGI M. Molecular cloning and transcriptional analysis of WRKY and solavetivone biosynthetic genes in the hairy roots of Hyoscyamus albus [J]. Plant Gene, 2016, 5: 78−86. DOI: 10.1016/j.plgene.2016.01.001
[18] 廖俊杰, 李文静, 李进进, 等. 铁皮石斛有效成分与功效研究进展 [J]. 广东轻工职业技术学院学报, 2015, 14(3):16−19, 26. DOI: 10.3969/j.issn.1672-1950.2015.03.004 LIAO J J, LI W J, LI J J, et al. Advances in the of constituents and pharmacological effects of Dendrobium candidum wall. ex. Lind [J]. Journal of Guangdong Industry Technical College, 2015, 14(3): 16−19, 26.(in Chinese) DOI: 10.3969/j.issn.1672-1950.2015.03.004
[19] 施仁潮, 竹剑平, 李明焱. 铁皮石斛抗肿瘤作用的研究进展 [J]. 中国药学杂志, 2013, 48(19):1641−1644. SHI R C, ZHU J P, LI M Y. Research progress of anti-tumor effect of Dendrobium officinale kimura et migo [J]. Chinese Pharmaceutical Journal, 2013, 48(19): 1641−1644.(in Chinese)
[20] 吕圭源, 颜美秋, 陈素红. 铁皮石斛功效相关药理作用研究进展 [J]. 中国中药杂志, 2013, 38(4):489−493. LYU G Y, YAN M Q, CHEN S H. Review of pharmacological activities of Dendrobium officinale based on traditional functions [J]. China Journal of Chinese Materia Medica, 2013, 38(4): 489−493.(in Chinese)
[21] 吴蓓丽, 吴月国, 赵铮蓉, 等. 铁皮石斛免疫调节作用及相关活性成分多糖的研究进展 [J]. 中草药, 2019, 50(21):5373−5379. DOI: 10.7501/j.issn.0253-2670.2019.21.034 WU B L, WU Y G, ZHAO Z R, et al. Advances in immunoregulatory effects and bioactive compounds of polysaccharides from Dendrobium officinale [J]. Chinese Traditional and Herbal Drugs, 2019, 50(21): 5373−5379.(in Chinese) DOI: 10.7501/j.issn.0253-2670.2019.21.034
[22] TAMURA K, STECHER G, PETERSON D, et al. MEGA6: molecular evolutionary genetics analysis version 6.0 [J]. Molecular Biology and Evolution, 2013, 30(12): 2725−2729. DOI: 10.1093/molbev/mst197
[23] 林江波, 邹晖, 王伟英, 等. 铁皮石斛DoLIS基因克隆与茉莉酸甲酯诱导表达分析 [J]. 福建农业学报, 2020, 35(10):1071−1077. LIN J B, ZOU H, WANG W Y, et al. Cloning and expressions of LIS in Dendrobium officinale [J]. Fujian Journal of Agricultural Sciences, 2020, 35(10): 1071−1077.(in Chinese)
[24] 宋展, 高鑫, 吴冕, 等. 细胞色素P450酶的结构、功能与应用研究进展 [J]. 微生物学通报, 2020, 47(7):2245−2254. SONG Z, GAO X, WU M, et al. Structure, function, and application of cytochrome P450 enzymes [J]. Microbiology China, 2020, 47(7): 2245−2254.(in Chinese)
[25] CHAPPLE C. Molecular-genetic analysis of plant cytochrome p450-dependent monooxygenases [J]. Annual Review of Plant Physiology and Plant Molecular Biology, 1998, 49: 311−343. DOI: 10.1146/annurev.arplant.49.1.311
[26] PEDRAS M S C, YAYA E E, GLAWISCHNIG E. The phytoalexins from cultivated and wild crucifers: Chemistry and biology [J]. Natural Product Reports, 2011, 28(8): 1381. DOI: 10.1039/c1np00020a
[27] AHUJA I, KISSEN R, BONES A M. Phytoalexins in defense against pathogens [J]. Trends in Plant Science, 2012, 17(2): 73−90. DOI: 10.1016/j.tplants.2011.11.002
[28] 李珂, 马良, 杜鹏飞, 等. 玉米萜类植保素代谢关键基因对小斑病侵染的防御响应分析 [J]. 西北植物学报, 2015, 35(9):1776−1780. DOI: 10.7606/j.issn.1000-4025.2015.09.1776 LI K, MA L, DU P F, et al. Gene expression of maize terpenoid phytoalexin metabolism in response to southern leaf blight [J]. Acta Botanica Boreali-Occidentalia Sinica, 2015, 35(9): 1776−1780.(in Chinese) DOI: 10.7606/j.issn.1000-4025.2015.09.1776




 下载:
下载: